Quality Policy
Summary
This business policy defines the commitment Resolian makes to placing Quality at the heart of the work we do, and sets out our approach to implementing a proactive, risk-based Quality Management System.
Scope
This business policy applies to all employees and all Resolian sites.
Business Policy
Regulatory compliance. Resolian will meet all the necessary compliance requirements of our customers and regulators in whichever region we operate. In particular, Resolian works in compliance with Good Laboratory Practice, Good Clinical Practice and current Good Manufacturing Practice regulations, as applicable to the services provided, and maintains the licenses and permits needed to operate.
As an analytical service provider, Resolian places Data Integrity at the heart of our Quality Management System (QMS).
Strategy. Resolian’s QMS is aligned to the needs of the business and supports our pillars and strategic imperatives:
- A positive Quality Culture is central to the way we work. Our Quality Culture is integrated into our talent development and Everyone in the organization must take responsibility for the Quality of their work.
- We take a risk-based approach to Quality and put the safety of patients above everything We adopt a Quality by Design approach and build quality into our processes and systems.
- We are client focused and seek to be easy to work with in everything we We strive to implement Quality in pragmatic, flexible and agile ways. We develop close relationships with our clients and understand their needs.
- Wherever possible, we operate an integrated and harmonized QMS and services across all our sites.
- We maintain our awareness of current and future Quality and Regulatory requirements, through industry and regulatory forums, so that we can anticipate and develop our QMS proactively.
Staff and training. We invest in our people through training and the development of their skills. Our regulatory compliance is built on an understanding of the underlying principles, purpose, and reasons for the requirements (“informed compliance”).
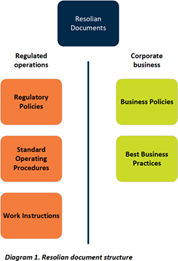
Documentation. Resolian controls our regulatory and corporate business documents in appropriate, separate, document management systems (see Diagram 1). Corporate documents do not describe regulatory requirements.
External audit and inspection. All policies, procedures and other documents are the intellectual property of the business. They will be made available to clients and 3rd parties in a secure time-limited and read-only format for authorized audits.
Facilities and equipment. Resolian will ensure suitable facilities and equipment are available to perform the work. These will be validated where required to meet the appropriate regulatory requirements.
As a global business Resolian adopts an international date format in all records: DD MMM YYYY (e.g. 25 APR 2023)
Suppliers. Resolian will approve suppliers according to the business needs and associated risk of the services or goods. Resolian expects all of our suppliers to follow our Code of Conduct, and promotes a diverse and sustainable supply chain.
Governance & reporting. Resolian maintains an internal independent audit program covering all relevant aspects of the work and facilities. Quality metrics are monitored and reported to local and corporate management functions and are used to drive continuous improvement across the business.